President Trump says the U.S. has Struck a Deal with Moderna to Receive 100 Million Doses of Coronavirus Vaccines

U.S. President Donald Trump announced on Tuesday that the U.S government has reached an agreement with Moderna (MRNA) to receive 100 million doses of experimental COVID-19 vaccine, which is already in the late-stage human trials phase.
In relation to the agreement, Moderna said the deal for its mRNA-1237 vaccine is worth $1.53 billion and will avail the federal government the opportunity of getting up to 400 million additional doses. So far, the U.S. government has invested $955 million in Moderna’s coronavirus vaccine development, bringing the total investment to $2.4 billion, according to the company.
The mRNA vaccine which is already in the late-stage human trials phase will conduct its final tests on 30,000 people. Moderna said it expects the results to be ready by October 2020.
The U.S. government also has ongoing deals with other pharmaceuticals like Pfizer and Johnson and Johnson for potential coronavirus vaccines. The federal government’s deals across these pharmaceuticals are similar to the agreements made with Moderna.
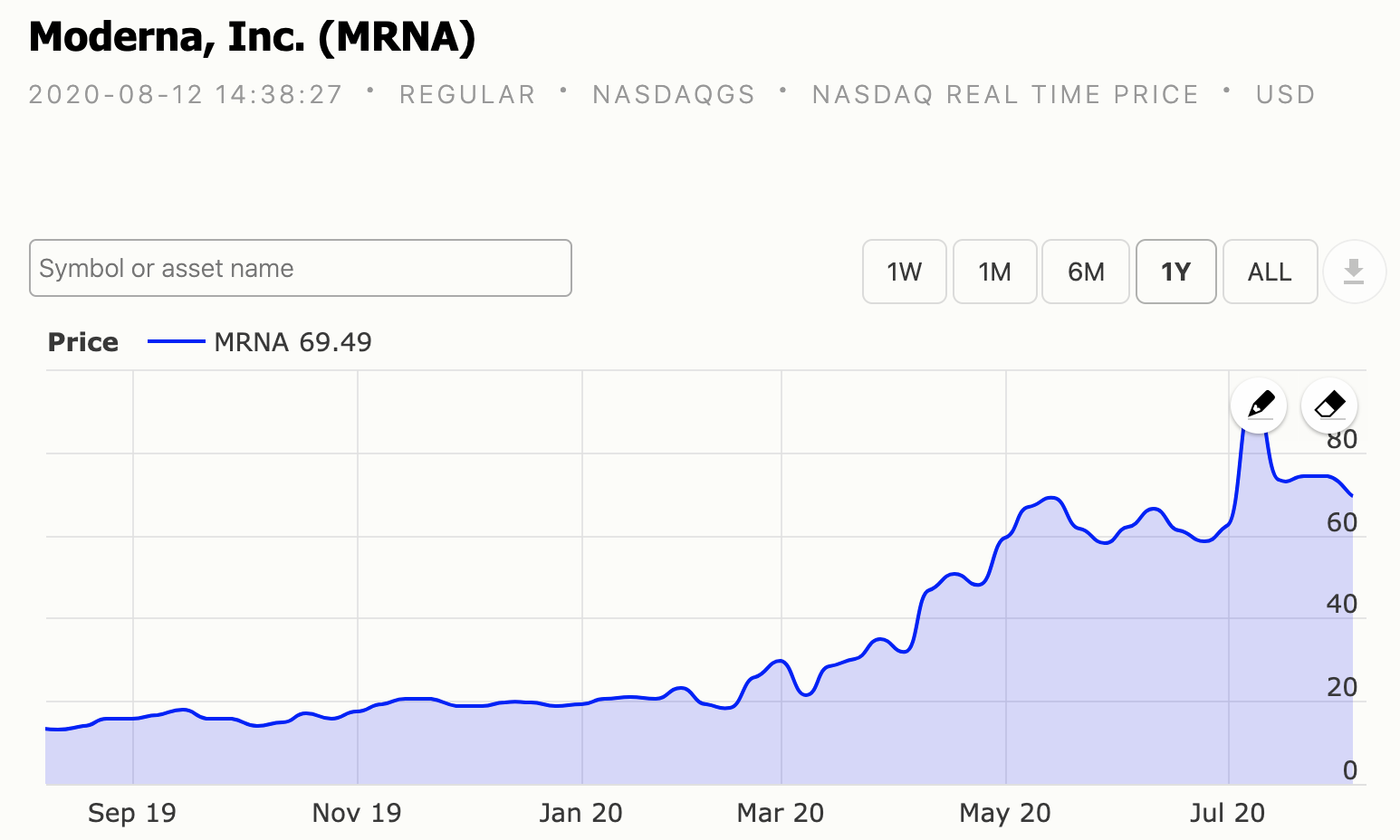
Moderna One year chart.
“We are investing in the development and manufacture of the top six vaccine candidates to ensure rapid delivery. The military is ready to go, they’re ready to deliver a vaccine to Americans as soon as one is fully approved by the FDA and we’re very close to that approval,” Trump said at a White House press conference.
The U.S. Department of Health and Human Services also previously said it would pay Pfizer and BioNTech a sum of $1.95 billion to produce and deliver 100 million doses of their coronavirus vaccine, with an option of purchasing additional 500 million doses. The HHS also announced that it would invest more than $1 billion in Johnson and Johnson to receive 100 million vaccine doses with the option of purchasing 200 million additional doses.
The Department of Health and Human Services also has ongoing deals with GlaxoSmithKline and Sanofi worth $2.1 billion to produce and deliver 100 million doses of their potential COVID-19 vaccine with the option of acquiring 500 million additional doses.
Both the U.S. government and the HHS deals are all part of the Trump administration’s efforts to boost the development, production, and delivery of COVID-19 vaccines and treatments to help curb the disease. All the COVID-19 related pharmaceutical deals fall under the Operation Warp Speed program.
“We appreciate the confidence of the U.S. government in our mRNA vaccine platform and the continued support,” said Moderna CEO Stephane Bancel, in a press release.
The mRNA-1237 vaccine was developed by the pharmaceutical Moderna in collaboration with the U.S. government. The vaccine received development assistance from the National Institute of Allergy and Infectious Disease, and the U.S. Biomedical Advanced Research and Development Authority (BARDA), which also supported the late-stage clinical trials and has helped boost production.


Be the first to comment!
You must login to comment